盲肠结扎穿孔以及电针刺激
1. SD大鼠适应性饲养一周
2. 水合氯醛麻醉,然后打开腹部,对盲肠用生锈的注射器针头刺穿
3. 术后缝合,观察动物状态,并对电针组实验动物进行电针治疗
4. 在6h、12h分别处死部分动物取血
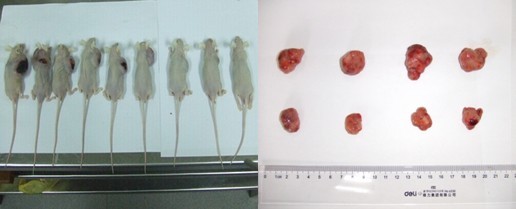
肿瘤模型(皮下种植结肠癌/肝癌/肺癌模型)
1. 购买4~6周的裸鼠,适应性饲养一周
2. 前肢腋下部位附近皮下种植结肠癌细胞,浓度1X107个/100ul
3. 注意无菌,然后饲养观察
4. 称重和实验观察和记录
5. 一般实验需要4周左右
大鼠心梗模型
1. 购买4-6周的大鼠,适应性饲养一周
2. 进行大鼠冠脉结扎,于末次给药后1h,在戊巴比妥钠腹腔麻醉下仰位固定于手术台,自左侧3-4肋间开胸,暴露心脏,于肺 动脉圆锥及左心房间找出冠脉左前降支,立即结扎冠脉,将心脏送回胸腔,并挤出胸腔内血液和气体,迅速关闭胸腔,开胸时间不超过30s。术后5天连续注射青霉素钠
3. 观察开胸后、缝针后、结扎后、关胸后心电图的变化
4. 合格后的大鼠在4周及6周后复查心电图
自体静脉移植
1. 选新西兰大耳兔,戊巴比妥钠按照体重麻醉
2. 切开颈部皮肤,钝性分离皮下组织;选取大小和直径同要移植部位的血管相同的静脉血管,先结扎该静脉的两端
3. 剪下该静脉,用灭菌生理盐水冲洗干净,备用
4. 将要移植部位的动脉血管,同周围组织分离,两端用动脉夹夹闭,然后剪下,把断端清洗干净;把前面的静脉缝合到该动脉断端。先稍微放开远心端的动脉夹,检查是否有渗血,确认无渗血后,同样 方法检查另外一端
5. 分层缝合,在关闭皮肤前,进行严格清洁和抗菌处理,然后缝合皮肤
6. 术后两周内静脉注射适量的肝素抗凝,随时观察实验动物的状态,及时处理;根据客户自己确定实验周期,准备动物和实验器材至少需要一周
贴剂动物体外实验
1. 实验分组及处理
2. 用药24h后取下,用温水洗脱
3. 分别于1h,24h,48h,72h肉眼观察局部有无红斑和水肿。然后连续用药7d,每天贴敷时间为12h,观察停药1h,24h,48h,72h后局部红斑、水肿情况,以及是否有色素沉着、出血点、皮肤粗糙或皮肤菲薄等情况,记录发生和消退时间
皮肤急性毒性试验
1. 实验分组及处理
2. 用药24h后取下,用温水洗脱
3. 分别于1h,24h,48h,72h至第7天,每天观察并记录动物的体重、皮毛光泽、饮食、眼和粘膜的变化,呼吸中枢、神经系统、四肢活动和死亡情况
4. 并于7天后处死大鼠,解剖观察其心、肝、肺、肾、卵巢、子宫等脏器有无异常变化,详细记录任何组织或器官出现的体积、颜色、质地的改变
5. 主要脏器称重,计算脏器指数
皮肤过敏试验
1. 实验分组及处理
2. 脱毛24h后,取痛经巴布贴贴敷在SD大鼠脱毛区,并以无刺激胶布固定,持续6h,第7天和第14天以同样方法重复一次
3. 于末次给痛经巴布贴巴布剂致敏后14d,将痛经巴布贴贴敷于SD大鼠脱毛区,赋形剂贴对照组与阳性对照组方法同上
4. 6h后去掉受试药物,即刻观察,并于24h,48h,72h再观察有无过敏反应,按表一记录个时间过敏反应分值,同时注意观察SD大鼠是否有哮喘、站立不稳或休克等严重的全身性过敏反应,计算致敏发生率
药效学实验
1. 实验分组及处理
2. 采用戊酸雌二醇+催产素引起大鼠子宫收缩为痛经模型。大鼠灌胃0.8mg/只/天(第1天和第10天),其它时间0.4mg/只/天,连续15天,第15天腹腔注射催产素2u/kg
3. 一般行为观察:精神状态、活动度、体重、食量、死亡率等
4. 扭体反应发生率观察:注射催产素后观察30min扭体反应发生率
5. 疗效观察:各组模型大鼠子宫、卵巢精确称重,计算子宫、卵巢指数
6. 观察对模型大鼠血液流变学的影响
IBD模型
1. 造模前 1d 禁食 24h 不禁水称重后,3%的戊巴比妥腹腔注射麻醉(或乙醚) 5%(W/V)的2,4,6-三硝基苯磺酸 (TNBS) 按 100mg/kg-150mg/kg(约等于TNBS 原液 0.4mL-0.6 mL,相当于 200 g 大鼠 20 mg-30 mg )
2. 分别加入乙醇,体位为头朝下,用聚丙烯管插入肛门上段4cm 注射器注入
3. 再倒立1-5min左右,以防药液溢出,每隔10d重复灌肠4次后可制成病鼠模型
4. 采血前观察SD大白鼠的情况,记录大鼠外观体征、行为活动、粪便的情况,记录死亡情况,每天称重
MTD实验
1. 动物称重并标记
2. 计算相应给药体积:按需计算每只动物的给药量,分为高中低3个梯度,并作记录
3. 给药操作
4. 给药后观察:在给药时记录给药时间。连续观察至少14天,观察一般指标(如动物外观、行为、对刺激的反应、分泌物、排泄物等)、动物死亡情况(死亡时间、濒死前反应等)、动物体重变化。记录所有的死亡情况、出现的症状,以及症状起始的时间、严重程度、持续时间等
肾脏缺血再灌注
1. 购买200g的SD大鼠,饲养一周,分组
2. 根据体重注射相应剂量的水合氯醛,麻醉后进行手术
3. 打开腹腔,钝性分离肾动脉,然后用动脉夹夹闭肾脏动脉30-45min,根据实验者的设计执行
4. 然后去除动脉夹,肾脏再次流入动脉血,肾脏由紫黑重新变为鲜红色
5. 然后根据实验要求取材
成年营养性肥胖大鼠模型
1. 购买体重200g的雄性SD大鼠
2. 方法 :用高脂 高糖饲料喂养成年大鼠四至八周
3. 观察大鼠体重及Lee’s指数、腹腔内脂肪重量及脂肪系数、葡萄糖耐量、血脂、血糖及血胰岛素水平 ,并算出胰岛素敏感性指数等
转基因小鼠筛选——鼠尾提取基因组DNA方法
1. 小鼠分笼后打耳标,剪取小鼠尾尖3mm-5mm左右于1.5mL 离心管中,标记耳标号
2. 配置消化液(SSTE),每管加入消化液0.5mL,蛋白酶K 1-5 μL, 55℃,3-5h,或放置过夜。(最长可放置3天)
3. 加1倍体积(0.5mL)PCI(下层液体),上下颠倒10余次。(PCI可分装出一些现用,以免操作不慎污染)
4. 室温15000rpm离心,10min
5. 取上清约0.4mL入新管,依次标号,加入异丙醇0.4mL ,4℃,15000rpm离心,5min
6. 弃上清,滤纸吸干,加入70%乙醇0.5mL,将管底沉淀弹起,上下颠倒几次,洗涤
7. 4℃,15000rpm离心,5-7min
8. 弃上清,瞬时离心,用枪吸干剩余液体,后经空气干燥5-10min
9. 加入1*TE 60-170μL一般加100μL,振荡30min溶解,于4℃冰箱保存
酶联生物经过不断的实验优化和改进,积累了大量的经验,拥有专业的酶联研发团队。利用专业的酶联免疫技术自主研发的elisa试剂盒,能对血清及其它样本定量检测抗原,定性检测特异性抗体。优质的试剂,先进的仪器和正确的操作是保证ELISA检测结果准确可靠的必要条件。ELISA检测的方便性、稳定性、重复性和可靠性方面都具有很大的优势。
ELISA检测技术服务内容:
1、双抗体夹心法检测抗原 2、间接法检测抗体 3、为客户提供各种ELISA技术进行样本检测。

以上代测费,凡购买本公司试剂盒,我们免费代测!
凡购买本公司目录任何一种酶联免疫检测试剂盒,您只需将需要检测的动物(Human, Rat, Mouse, Rabbit, Monkey,
Pig……)种类和检测指标(白介素类、激素类)及标本数量(48T/96T)通知公司业务员即可。在接到客户标本当日起,现货产品一周内将检测报告交到客户手中!
欢迎各科研单位在各种项目上与我们公司开展不同层次的密切合作,以双赢求发展,共同进步,为中国检测事业的发展积累经验。
二、样本要求
在收集标本前都必须有一个完整的计划,必须清楚要检测的成份是否足够稳定。我们提倡新鲜标本尽早检测,对收集后当天就进行检测的标本,及时储存在4℃备用,如有特殊原因需要周期收集标本,请造模取材后,将标本及时分装后放在-20℃或-70℃条件下保存。因冰室与室温存在一定温差,蛋白极易降解,直接影响实验质量,所以避免反复冻融。代测放免标本的客户取材前须向我司销售人员索要说明书,具体操作注意事项请与我司技术人员沟通。
液体类标本:标本必须为液体,不含沉淀。包括血清、血浆、尿液、胸腹水、脑脊液、细胞培养上清、组织匀浆等。
血清:室温血液自然凝固10-20分钟后,离心20分钟左右(2000-3000转/分)。收集上清。如有沉淀形成,应再次离心。
血浆:应根据试剂盒的要求选择EDTA、柠檬酸钠或肝素作为抗凝剂,加入10%(v/v)抗凝剂(0.1M柠檬酸钠或1%heparin
或2.0%EDTA.Na2)混合10-20分钟后,离心20分钟左右(2000-3000转/分)。仔细收集上清。如有沉淀形成,应再次离心。
尿液、胸腹水、脑脊液:用无菌管收集。离心20分钟左右(2000-3000转/分)。仔细收集上清。如有沉淀形成,应再次离心。
细胞培养上清:检测分泌性的成份时,用无菌管收集。离心20分钟左右(2000-3000转/分)。仔细收集上清。检测细胞内的成份时,用PBS(PH7.0-7.4)稀释细胞悬液,细胞浓度达到100万/ml左右。通过反复冻融,以使细胞破坏并放出细胞内成份。离心20分钟左右(2000-3000转/分)。仔细收集上清。保存过程中如有沉淀形成,应再次离心。
组织标本:切割标本后,称取重量。加入一定量的PBS,缓冲液中可加入1μg/L蛋白酶抑制剂或50U/ml的Aprotinin(抑肽酶)。用手工或匀浆器将标本匀浆充分。离心20分钟左右(2000-3000转/分)。仔细收集上清置于-20度或-70度保存,如有必要,可以将样品浓缩干燥。分装后一份待检测,其余冷冻备用。
三、寄标本时需注明以下情况:
1、标本编号;2、所测项目;3、是否做复孔;3、联系方式;4、实验后标本是否寄回。
客户须知:
客户应对所提供的材料及信息负责,如因客户提供的材料及信息不准确而引起的实验延误或经济损失由客户承担。
Q:1.
how to collect samples and preparation of ELISA?
Performed by ELISA test is generally common clinical samples including blood (finger
blood, blood), urine, feces, cerebrospinal fluid, pleural effusion, prostatic fluid,
semen, vaginal secretions, which
Some time of sample collection, preservation methods and has certain requirements.
Collection (a) clinical specimens
A, blood samples:Some physiological factors, such as smoking, eating, exercise, mood
swings, pregnancy, postural changes in blood can affect certain ingredients, even some
of diurnal variation. Therefore, blood samples
Acquisition should avoid interference physiological factors, consistent with appropriate
conditions, such as can not be avoided, should indicate the factors on the specimen.
1. Peripheral:Usually select the inside of blood left ring finger, the portion should be
no frostbite, inflammation, edema, damage. If the site does not meet the requirements to
other parts of the fingers instead. For burn patients, optional leather
Intact skin at the blood. As part of routine blood tests (eg, white blood cell count,
sort, etc.) affected by physiological factors fluctuation is too large, when compared to
the conditional should be consistent. It relates to the body, blood clotting function
Can test items (such as platelet count, bleeding time or clotting time) testing, we must
pay attention to understand whether the patient used anticoagulant, procoagulant drugs
in order to reduce or avoid interfering factors
influences.
2. Blood:In addition to involving a variety of projects such as hemostasis and
thrombosis detector requires the use of anticoagulated blood plasma, the current
analysis to detect the vast majority of projects can be directly detected using blood
serum. In the serum test items
, Some (such as blood sugar, blood fat) diet and circadian factors influenced, fasting
blood samples were generally appropriate; some decay rapidly in the blood (serum enzyme
activity assay such as ACP activity, etc.),
0 ~ 4 ℃ storage is not an activity decreased, the detection of these projects must be
timely and fast; some (such as creatine kinase) influenced by exercise and other
factors. Avoid hemolysis occurs when blood is also important
And, more particularly potassium, LDH and other measurement.
B, urine samples:With the same blood samples, urine samples affect diet, exercise,
medication and other factors that are also large, especially on the diet, so the morning
urine generally superior to random urine. Means getting up early morning urine
After the first urine specimens, representing concentrated and acidified visible
components (such as blood cells, epithelial cells, tubular) easy to observe the relative
concentration. Random urine that is a random urine specimens convenient, but by diet,
Sports, and even more the influence of drugs, prone to false positive and false negative
results, such as diet proteinuria, glucosuria diet, vitamin C interference occult blood
results and the like. Postprandial urine (patient 2 hours after lunch, collected
Human Urine) suitable for urine, urine protein and urobilinogen check urine samples at
this time to increase the sensitivity of the test, the detection of minor lesions. 12
hours in urine cell count is Addis count (last night 8:00
After emptying the bladder to all specimens of urine 8 o'clock the next morning),
because a long time, easy to breed bacteria shall be added preservative formaldehyde.
24-hour urine (the first day of the morning after emptying the bladder specimens from
8:00 to 8:00 the next morning
All urine) quantification of chemical substances, including proteins, sugars, urinary
17-one, 17-hydroxy steroids, catecholamines, Ca2 +, etc., to detect different
substances, choose a different preservative preservative. clean
Urine used for urine bacterial culture requires sterile specimens were taken after
washing the vulva. Urine specimens should be enough to collect all, at least 12 ml,
preferably 50 ml, the timing must collect all the urine of women
Patients should avoid vaginal secretions, blood contamination of urine specimens.
C, stool samples:Stool samples for the detection judgment digestive diseases has
important reference value. Collection requirements with a clean bamboo select faecal
mucus, pus and blood components and other abnormality, no abnormal appearance
Droppings shall be drawn from multiple surface and deep manure end. Get parasitemia and
for egg counts should be collected 24 hours feces. Dysentery amoeba trophozoites check
should immediately check in after a bowel movement, and from there sepsis
Softer at the drawn, insulation inspection. Charles S. japonicum eggs should take mucus,
pus and blood portion 30g stool specimens from at least miracidia hatching, and to be
treated as soon as possible. Check pinworm eggs must use transparent film swab
Night before 12:00 or early in the morning from defecation wrinkled folds around the
anus and immediately swabbing at microscopic examination. Occult blood test (chemistry),
fasting before the test on the 3rd of meat and foods containing animal blood and ban
clothing iron, vitamin C and so on.
Should be checked in all 1 hour stool specimen collection is completed, in order to
prevent damage to physical components of digestive enzymes and pH by. For clinical
samples above the detection indicators.
D, CSF samples:CSF samples collected immediately after submission, place too long will
affect the test results: such as cell degeneration, destruction, leading to counting and
classification are not allowed; some chemicals such as glucose content will decompose
Save
Less; bacteria occur autolysis affect bacteria detection rate. Cerebrospinal fluid
extracted three general dispensing a sterile tube, the first tube for bacterial culture,
a second tube for chemical analysis and immunological tests, the third tube for general
Characters and microscopic examination, three of the order should be reversed. Specimen
collection is difficult because all inspection and testing process should pay attention
to safety.
E, ascites and pleural effusion samples:CSF samples with the same attention to safety
after the specimen collection, and timely submission. Generally separated into three
tubes, one for routine cytology, a biochemical examination, a bacterial culture, in
order
CSF same is appropriate.
F, prostatic fluid sample:Prostatic fluid specimen after prostate massage by the
acquisition, directly drop when less liquid on a glass slide and timely submission shall
be taken to prevent sample evaporation to dryness, the amount collected for a long time
in a clean, dry test tube. If massage
No prostatic fluid, urine sediment can be checked after the massage.
G, semen samples:Abstinence before semen collection should be 3 to 7 days, drain the
urine after masturbation or other available methods of semen directly into clean
containers, insulation and timely submission. Due to changes in sperm production during
the day and
Large, generally should be checked 2 to 3 times (each time interval of 1 to 2 weeks) in
order to make a diagnosis.
H, samples of vaginal secretions:Vaginal samples were collected 24 hours before
intercourse should be prohibited, bath, vaginal examination, vaginal lavage and local on
the drug, etc., drawing instruments used need to be cleaned. Usually with brine-soaked
cotton swab from the vagina deep
Or rear vaginal fornix, cervical canal mouth drawn, etc., made after saline smear
vaginal secretion samples observation, women with menstrual vaginal secretions were not
checking.
2, do before each sample by ELISA experiment how to prepare?
Before collecting the sample must have a comprehensive plan must clearly be detected
component is stable enough. To be collected on the same day
Sample testing, and timely backup stored at 4 ℃. For the next day re-testing samples
frozen in a timely manner after dispensing -20 ℃ spare, conditional, preferably -70 ℃
cryopreservation standby. Avoid repeated freezing and thawing specimens
.
Liquid samples: including serum, plasma, urine, pleural effusion, cerebrospinal fluid,
cell culture supernatant and the like.
1. serum:
Coagulation at room temperature 10-20 mins, centrifugation 20 minutes or so (2000-3000
rev / min). Carefully collect the supernatant. If precipitation during storage,
Centrifugal again.
2. Plasma:
EDTA should be selected according to the requirements of the specimen, sodium citrate or
heparin as an anticoagulant, mix 10-20 mins, centrifugation 20 minutes or so (2000-3000
rev / min). Carefully collect the supernatant. Save process
If precipitation appeared, Centrifugal again.
3. Urine:
Sterile collection tube. Centrifuged for 20 minutes or so (2000-3000 rev / min).
Carefully collect the supernatant. If precipitation during storage, Centrifugal again.
Pleural and peritoneal effusions, and cerebrospinal fluid Reference to this practice.
4. The cell culture supernatant:
The detection of secretory component with a sterile collection tube. Centrifuged for 20
minutes or so (2000-3000 rev / min). Carefully collect the supernatant.
5. cultured cells
????When the detection of intracellular components, diluted with PBS (PH7.2-7.4) cell
suspension, the cell concentration reached 1 million / ml or so. By repeated freezing
and thawing or tissue protein extraction reagent was added to the cells
Damage and release of intracellular components. Centrifuged for 20 minutes or so
(2000-3000 rev / min). Carefully collect the supernatant. If precipitation during
storage, Centrifugal again.
6. tissues
????After cutting samples, check the weight. Adding a certain amount of PBS, PH7.4.
Rapidly frozen with liquid nitrogen. After thawing samples remained at 2-8 ℃. Adding a
certain amount of PBS
(PH7.4), or tissue protein extraction reagent, or by hand homogenizer homogenized
sample. Centrifuged for 20 minutes or so (2000-3000 rev / min). Carefully collect the
supernatant. A new package to be detected, which
I alternate freezing.
Q:Do
I have to run all of my standards and samples in duplicate?
A:Yes, the duplicates are run in order to monitor assay precision and increase
confidence in the assay results obtained.
Q:Do
I have to run all of my samples at one time?
A:No, each kit uses stripwell microplate. This allows the user to analyse different
numbers of samples at different times.
Q:What
types of reproducible results are obtained with the assays?
A:Each kit comes with a manual containing a graph of typical data obtained. Any
variation in operator, pipetting and washing technique, incubation time or temperature,
and kit age can cause variation in result. Each user should obtain their own standard
curve.
Q:Is
it possible to store the reagents other than indicated?
A:Storage of the kit components under conditions other than indicated is not recommended
in order to assure proper performance of the test.
Q:How
should I store my samples?
A:Samples should be stored at -20oC or lower temperature. For long-term storage, it is
recommended to freeze them at -70oC -80oC.
Q:Can
I modify the protocol?
A:BG ELISA kits have been optimized to provide the best possible results. Modifying the
format or protocol may give inaccurate and wrong results.
Q:Can
I use a sample type that is not recommended in the kit insert?
A:The kit has been validated for the sample types listed in the kit insert. Sample types
other than those validated have not been tested. Contact Technical Service for further
information.
Q:My
samples generated values that were outside the dynamic range of the assay. Can I use
these values?
A:It is recommended that only sample values that fall within the range of the standard
curve be used. Values outside the range of the standard curve are generally non-linear,
which can lead to incorrectly extrapolated values. Samples that generate values higher
than the highest standard should be (further) diluted and the assay repeated. If samples
fall below the range of the assay, the sample is considered to be non-detectable.
Q:Do
I have to run a Blank or Zero Standards every time?
A:Yes, these are required for the calculations, and reflect any subtle but significant
performance changes from day to day and assay to assay. They are also extremely helpful
when troubleshooting the source of a particular assay problem.
Q:Can
I alter the volume of sample I use in the assay?
A:It is not recommended that you alter the volumes since all BG kits are designed for
optimal performance at the given volumes
Q:Can
components from different kits be used?
A:Each kit contains components which have specific lot numbers to ensure that all of the
components are performing optimally alone, as well as with all of the other components
in the kit. QC testing is performed on these specific lots. It is never recommended to
use your own components or components from other kits or vendors.
Q:My
standard curve looked fine, but I didn’t get a signal in my sample when I expected
to, why?
A:The sample may not contain the analyte. A matrix effect may be masking the detection.
Ensure that the recommended dilution was followed as stated in the kit insert. If
dilution was recommended, check to be sure that the dilution was performed properly.
Over-dilution may cause the sample to fall below the range of the standard curve.
Q:How
do you recommend I wash my plate?
A:If you are using an automated plate washer we recommend that the calibration be
checked on a regular basis, and that the system is flushed with the Plate Washing Buffer
prior to washing. The same is true for a manual washer. A repeater or a wash bottle can
also be used. The user should be careful to ensure that all of the contents are
aspirated and the plate tapped dry on lint-free paper.
Q:Do
I need to use a plate shaker?
A:Reliable results can be obtained without a plate shaker, but the O.D.'s will generally
be lower than those obtained using a plate shaker.
Q:Why
do I have to use wavelength correction between 450-570nm?
A:For the ELISA assay, reading at dual wavelengths is done to correct for the optical
density contributed by the plastic well, the lamp and optical fluctuations.
Q:If
I extract my sample, do I still need to follow the recommended dilutions given in
the kit insert?
A:The amount of sample dilution needed after an extraction procedure will be affected by
the effects of purification and concentration in the protocol used. The amount of
dilution or concentration will have to be determined by the end-user.
Q:What
is the expected concentration of analyte that I should expect to find?
A:The amount of a given analyte may vary not only from species-to-species, but also
between tissue and cellular sources. The best source of this information is the current
literature that is easily accessed through the Internet at multiple scientific
databases.
Q:My
optical densities were a little higher (or lower) than those in the manual that came
with my kit. Why?
A:The optical density is affected by a number of physical conditions such as time and
temperature. We suggest that you shorten or lengthen the final incubation with substrate
solution to compensate.
Q:What
are the reasons for High Background?
A:1) Improper Washing: Check volume of washing buffer reservoir and make sure all
recommended washing steps are performed.
2) Contaminated Substrate: Make sure there is no contamination of the substrate with
metal ions or oxidizing reagents, before use. Keep the extra substrate solution
separately during the ELISA substrate development time.
3) Substrate exposed to light: Exposure to light may result in a blue color of the
substrate. Keep solutions in the dark (vial) until ready to dispense into the plate.
4) Wrong Incubation Times/Temperatures: Generally follow the test protocol regarding
incubation times and temperatures. However, if all wells are intensely and equally
colored with no intensity gradient observed in the standard dilution series, then it may
be necessary to observe the substrate reaction as the color is developing, in order to
stop the reaction sooner.


 酶联官方手机二维码
酶联官方手机二维码